
Global Health in the Age of AI
This whitepaper discusses the implications of AI on global health inequalities and proposes a coordinated approach to address these challenges.
Curated regulatory guidance, clinical analyses, and market reports. AI-summarized, editor-reviewed, instantly downloadable.

This whitepaper discusses the implications of AI on global health inequalities and proposes a coordinated approach to address these challenges.

This whitepaper discusses an innovative inline buffer formulation system that optimizes biomanufacturing processes.

This presentation discusses the validity of external controls in medical product development and their comparison to randomized controls.
This whitepaper analyzes the FDA's draft guidance on AI in drug development, detailing its implications for industry practices and regulatory compliance.

This paper presents a comprehensive dataset of digital therapeutic device approvals across four countries to aid regulatory decisions and industry analysis.

This report outlines the current status, trends, and regulatory challenges in the field of radiopharmaceuticals in the EU.

This whitepaper discusses the role of cytological specimens in diagnosing metastatic melanoma and their implications for precision oncology.
 🔒
🔒 A comprehensive catalog of professional policy positions by ASHP, advocating for pharmacy practice and public health.
 🔒
🔒 This whitepaper outlines the Medicare Advantage capitation rates and payment policies for 2027, emphasizing stakeholder feedback and risk adjustment strategies.
 🔒
🔒 This review discusses the 2024 revision of EU GMP Annex 1, focusing on contamination control and quality risk management in sterile manufacturing.
 🔒
🔒 This white paper outlines the scientific standards for 23andMe's genetic health and trait reports, emphasizing validity and evidence-based practices.

This position paper outlines AESGP's recommendations on simplifying medical device regulations in Europe.

This whitepaper discusses strategies for enhancing post-approval compliance in pharmaceuticals through integrated governance and AI.
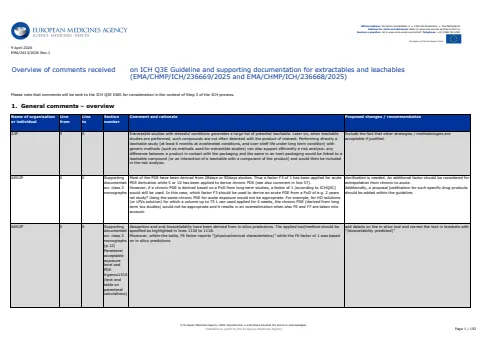
This document summarizes comments on the ICH Q3E Guideline regarding extractables and leachables, focusing on regulatory and safety considerations.

This whitepaper reviews the FDA's Accelerated Approval pathway, highlighting its evolution, challenges, and proposed reforms.