 🔒 ★
🔒 ★ European Medicines Agency Pre-Authorisation Procedural Advice
Guidance on procedural advice for users of the centralised procedure by the European Medicines Agency.
Curated regulatory guidance, clinical analyses, and market reports. AI-summarized, editor-reviewed, instantly downloadable.
 🔒 ★
🔒 ★ Guidance on procedural advice for users of the centralised procedure by the European Medicines Agency.
 🔒 ★
🔒 ★ Guidance on procedural advice for users of the centralised procedure by the European Medicines Agency.

This consensus document outlines strategies to increase female representation in cardiovascular clinical trials, addressing significant under-representation of women.

The updated BSG guidelines emphasize the necessity of ileocolonoscopy for IBD diagnosis and advocate for immunomodulators in all patients.

This study identifies key barriers and facilitators impacting healthcare practitioners' use of digital health technologies during and before the COVID-19 pandemic.
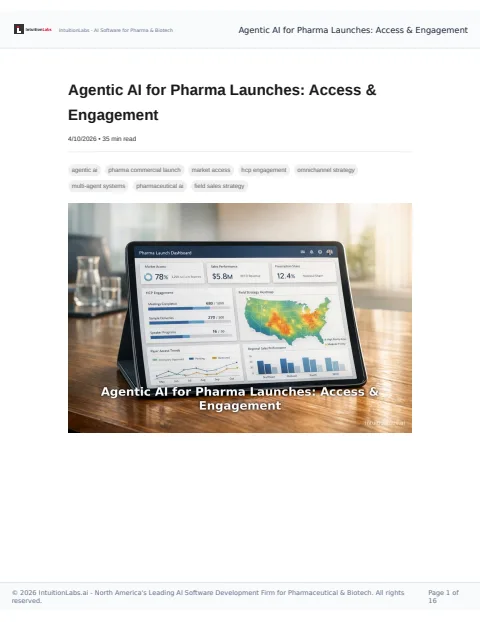 🔒
🔒 Agentic AI can enhance pharma launches by improving market access and HCP engagement, with potential efficiency gains of up to 80%.
 🔒
🔒 The rise of GLP-1 agonists like Semaglutide demands innovative ready-to-use cartridges to enhance delivery efficiency and reduce costs in obesity treatment.
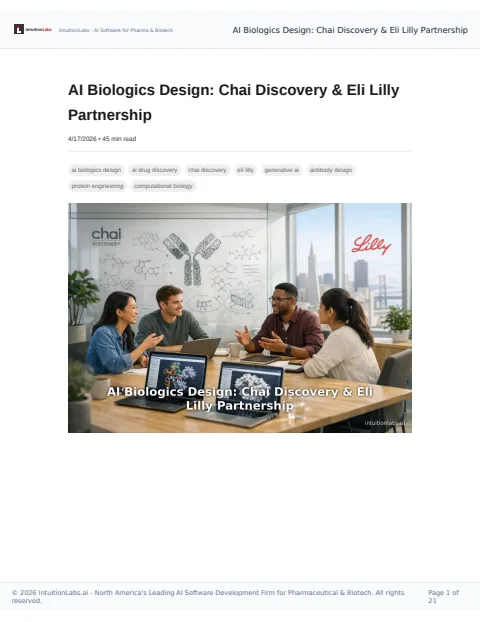 🔒
🔒 Eli Lilly's partnership with Chai Discovery aims to revolutionize biologic drug design using AI, potentially achieving hit rates of 20% in antibody development.
 🔒
🔒 Cigna's updated policy outlines conditions for acupuncture coverage, including migraine and musculoskeletal pain, effective April 2026.
 🔒
🔒 Effective patient mastering is crucial for healthcare organizations to ensure accurate patient journeys and reliable analytics, impacting strategic decisions and financial outcomes.
 🔒
🔒 At-risk Medicare Advantage arrangements show higher quality and efficiency compared to fee-for-service models, impacting care delivery for beneficiaries.
 🔒
🔒 This whitepaper outlines how MedTech can leverage automotive software development practices to enhance compliance and accelerate innovation cycles.
 🔒
🔒 Eli Lilly's daily weight-loss pill received FDA approval, setting the stage for a competitive battle with Novo Nordisk's Wegovy in the $70 billion GLP-1 market.
 🔒
🔒 This policy outlines medically necessary radiation therapy protocols for conditions like breast cancer and lung cancer, effective April 1, 2026.
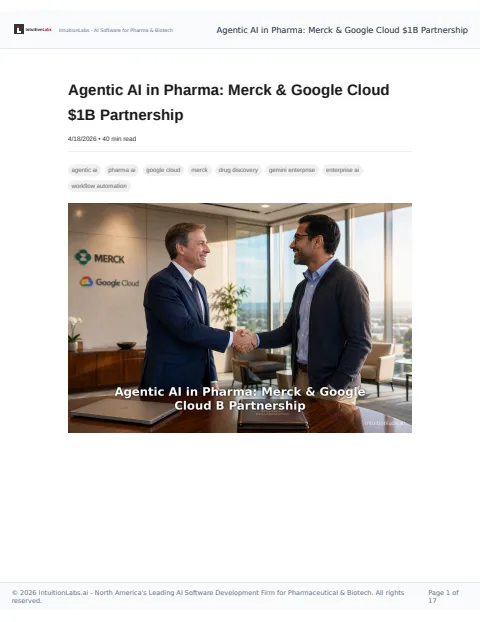 🔒
🔒 Merck's $1 billion partnership with Google Cloud aims to deploy agentic AI to transform drug discovery and boost productivity across its global enterprise.
 🔒
🔒 This whitepaper analyzes the market benchmark role in negotiating oncology drug prices in Italy, highlighting key implications for clinical practice.
 🔒
🔒 Pharma faces a $5B crisis as 35-40% of patients abandon specialty prescriptions. Misinformation from social media is eroding treatment adherence.

This whitepaper outlines the clinical appropriateness guidelines for chest imaging, aimed at improving patient care and standardizing practices.

This medical policy outlines guidelines for urine drug testing in pain management and substance use treatment.