 🔒 ★
🔒 ★ European Medicines Agency Pre-Authorisation Procedural Advice
Guidance on procedural advice for users of the centralised procedure by the European Medicines Agency.
Curated regulatory guidance, clinical analyses, and market reports. AI-summarized, editor-reviewed, instantly downloadable.
 🔒 ★
🔒 ★ Guidance on procedural advice for users of the centralised procedure by the European Medicines Agency.

This presentation discusses the validity of external controls in medical product development and their comparison to randomized controls.

This report outlines the current status, trends, and regulatory challenges in the field of radiopharmaceuticals in the EU.
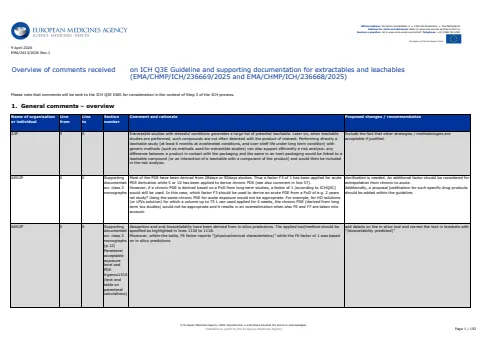
This document summarizes comments on the ICH Q3E Guideline regarding extractables and leachables, focusing on regulatory and safety considerations.