Press Releases
Page 9 • 12 itemsAccess critical pharma & biotech press releases. Stay ahead on drug approvals, clinical trials, M&A, and regulatory updates impacting global markets.

Celerion Secures Majority Investment from THL Partners to Expand Clinical Research Services
Clinical research organization Celerion announces majority investment from THL Partners to accelerate growth in drug development services and bioanalytical capabilities.

Veristat Acquires Certara's Regulatory Writing Business to Expand European Market Services
Veristat announces acquisition of Certara's regulatory and medical writing division to enhance clinical trial services for companies entering European markets.

Lung Cancer Therapeutics Market to Hit $142.3 Billion by 2036 as Precision Medicine Drives 11.8% Growth
Global lung cancer therapeutics market projected to grow from $46.8B to $142.3B by 2036, driven by precision medicine and immunotherapy advances.

Drug Delivery Devices Market Poised for Significant Growth Through 2032 Driven by Biologics and Regulatory Support
Drug delivery devices market shows strong growth potential through 2032, fueled by increasing biologics use, technological advances, and supportive regulations.

Polycythemia Vera Market Transformation: 9 Companies Including Protagonist Therapeutics with Rusfertide to Enter by 2036
Nine pharmaceutical companies are advancing polycythemia vera treatments through clinical trials, with Protagonist Therapeutics' rusfertide leading market expansion by 2036.

Ipsen's Ojemda (Tovorafenib) Becomes First Targeted Therapy for Pediatric Brain Tumors in EU
Ipsen's Ojemda receives EU approval as first targeted therapy for pediatric low-grade glioma, addressing critical unmet need in rare brain tumors.

Sanofi's Tzield Receives FDA Approval for Children as Young as 1 Year to Delay Type 1 Diabetes Progression
FDA approves Sanofi's Tzield for children aged 1+ with stage 2 type 1 diabetes, marking the first disease-modifying therapy for this pediatric population.

Kelun-Biotech's Sac-TMT Cancer Drug Selected for Three ASCO 2026 Oral Presentations
Kelun-Biotech announces three clinical studies of sac-TMT selected for oral presentations at ASCO 2026, highlighting progress in cancer treatment.
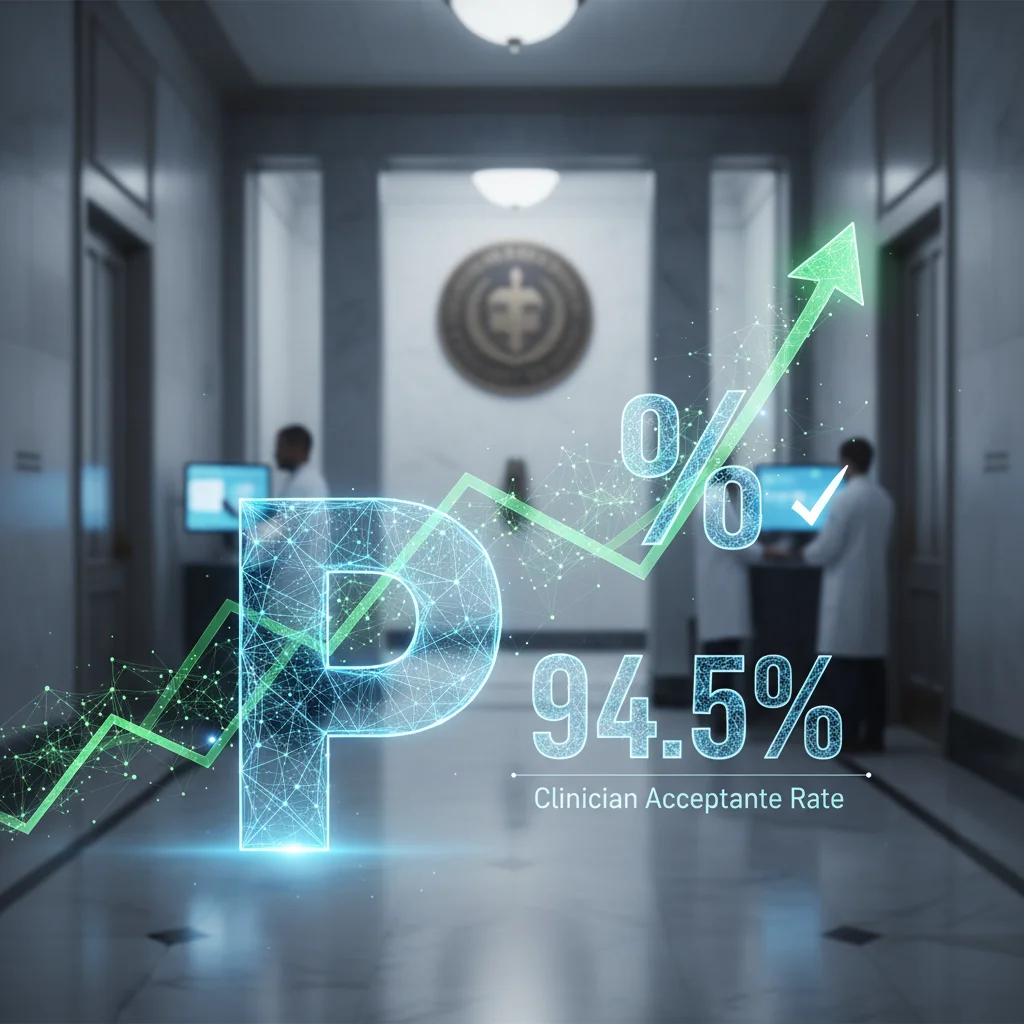
PrescriberPoint AI Prior Authorization Platform Achieves 94.5% Clinician Acceptance Rate in Clinical Study
PrescriberPoint's AI-powered prior authorization solution automates the full PA lifecycle with 94.5% clinician acceptance, streamlining prescription approvals.

GRAIL's Galleri Cancer Detection Test Shows Major Results in 174,000-Patient Studies at ASCO 2026
GRAIL presents breakthrough NHS-Galleri and PATHFINDER 2 trial data for multi-cancer early detection test at ASCO 2026, covering 174,000+ participants.

Eupraxia Pharmaceuticals Reports Positive 36-Week Data from Highest Dose Cohort in Eosinophilic Esophagitis RESOLVE Trial
Eupraxia's highest dose cohort shows robust tissue health and symptom improvements at 36 weeks in Phase 1b/2a eosinophilic esophagitis trial.

CellCarta Saves 9 Hours Weekly Using RegASK AI Platform for Regulatory Compliance
CellCarta eliminates regulatory bottlenecks with RegASK's AI platform, saving 9 hours per week and streamlining pharmaceutical compliance processes.
